Draw a Labelled Diagram of Antibody Molecule
What are antibodies?
Antibodies are specialized, Y-shaped proteins that bind like a lock-and-key to the body'southward foreign invaders — whether they are viruses, bacteria, fungi or parasites. They are the "search" battalion of the allowed system'south search-and-destroy organization, tasked with finding an enemy and marking it for destruction.
"They're released from the jail cell and they leave and chase," said Dr. Warner Greene, the director of the Centre for HIV Cure Inquiry at the Gladstone Institutes in San Francisco.
When antibodies find their target, they bind to it, which then triggers a cascade of actions that shell the invader. Antibodies are part of the so-chosen "adaptive" immune organisation, the arm of the immune organization that learns to recognize and eliminate specific pathogens, Greene said.
Related: Diagram of the human allowed system (infographic)
What do antibodies look similar?
The 2 arms at the top of the antibody's Y shape bind to what's known equally the antigen. The antigen can be a molecule, or a molecular fragment — often some part of a virus or leaner. (For instance, the new coronavirus SARS-CoV-2 has unique "spikes" on its outer coat, and some antibodies bind to and recognize these fasten proteins.)
The lesser of the Y, or the stem, binds to several other immune-organisation compounds that tin can assist kill the antigen or mobilize the immune system in other ways. Ane set up of these, for instance, triggers the complement pour, Greene told Live Scientific discipline.
"Complement is really the executioner," that punches holes in the target jail cell, such as the membrane of a virus, Greene said.
Antibodies, which are also called immunoglobulins (Ig), all take the same bones Y-shape, but at that place are five variations on this theme — called IgG, IgM, IgA, IgD and IgE, said Jason Cyster, a professor of microbiology and immunology at the University of California, San Francisco.
Each variation looks slightly different and plays slightly different roles in the allowed arrangement. For instance, immunoglobulin G, or IgG, is just one Y, whereas IgM looks a bit like the 10-armed Hindu goddess Durga, with 5 Ys stacked together, and each prong tin bind one antigen.
IgG and IgM are the antibodies that broadcast in the bloodstream and get into solid organs, Cyster said. IgA is "squirted out of the body," in fungus or secretions, Cyster told Live Science. IgE is the antibody that typically triggers allergic responses, such equally to pollen or peanuts, according to the American University of Allergy, Asthma & Immunology. IgD has historically been enigmatic, simply one of its roles is to help activate the cells that brand antibodies.
Related: Hay fever & seasonal allergies: Symptoms, causes & treatment

Where do antibodies course?
To understand antibodies, you beginning need to know about B-cells, which are a type of white blood cell that forms in the bone marrow. There are about a trillion B-cells in the body, and each ane has a unique IgM antibody that sits on the B-jail cell surface and each binds, to one antigen, said Simon Goodman, the Science and Technology Plan Manager for The Antibody Social club, a nonprofit organization that represents those involved in antibody inquiry and development.
This staggering level of variation allows the body to recognize almost whatsoever substance that could enter. Hither'due south how it achieves that diverseness: In each B-jail cell, the genes that lawmaking for the antibody's binding site are shuffled like playing cards in a deck.
"The amount of rearrangement that can occur is enormous," Cyster told Live Science.
Related: xi surprising facts about the immune system
These B-cells then patrol the body, often lingering longer in areas like the lymph nodes or the tonsils, Cyster said. Near of the time, these B-cells don't bind anything. But if, by a ane- in-a-meg chance, a B-prison cell does bind some foreign substance, "that triggers the B-cell to say 'Hey nosotros need to get activated,'" Cyster said.
The B-prison cell grows in size and starts to carve up in what'due south called "clonal expansion," Cyster said.
"It's an identical copy of the parent, but like the female parent," Cyster said. Later on a week or so, there may be hundreds of thousands to a million of these copies.
Eventually, these clonally expanded B-cells differentiate into plasma cells, which are antibody factories.
"They secrete x,000 antibodies per prison cell per 2d. They tin do that for weeks or years if y'all're lucky," Cyster said.
But not all B-cells carve up the aforementioned amount.
"If yous consider the B-jail cell to be a lock, and you consider all of these unlike things to exist floating around to be different keys, then some of the keys will fit better, some volition fit worse, and some won't fit at all," Goodman told Live Science. "And depending on how well the key fits into the lock on the surface of a particular B-jail cell, that cell will be triggered to divide more." Then, the more prolific B-cells produce more plasma cells and churn out more of a specific type of antibiotic.
The trunk doesn't just produce one type of antibody either; it produces a messy, chaotic zoo of them. Each locks onto different parts of an invader.
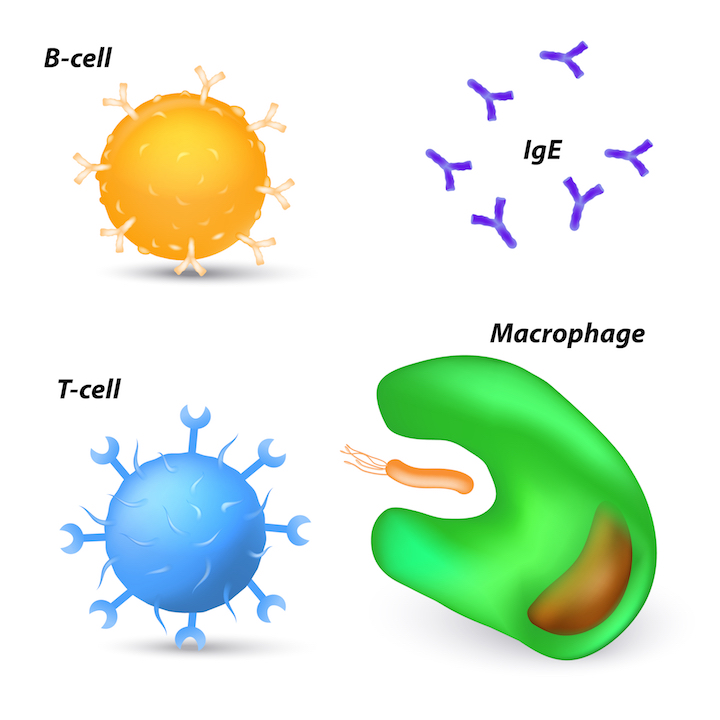
And antibodies don't all exercise the same thing once they've spring to a target. Some volition nip infection in the bud past direct neutralizing a threat, preventing a pathogen from entering a jail cell. Others tag invaders, so that the immune system's killer cells (which aren't antibodies) can remove it, Greene said. Still others may wrap viruses or bacteria in a gooey coating. And other antibodies might tell Pac-Human-like allowed cells called macrophages to come gobble upward the invader. (That strategy can sometimes backfire with viruses, which may co-opt this response to invade new cells, Cyster added.)
The first blazon of antibiotic to course afterwards you lot are exposed to a virus is IgM, which emerges within 7 to ten days later exposure, Greene said. IgM can bind to an invader, simply each "Y" in this 10-armed poly peptide does so fairly weakly. But, just as five weak people working together tin tackle a big, strong adversary, IgM's v Y'due south (10 arms) working together can bind tightly to an antigen, he added.
At well-nigh x to 14 days, the body begins making IgG, which is the immune system's "major workhorse," Greene said. IgG can cantankerous the placenta in a significant woman, giving a newborn passive protection against disease until their own immune organisation can ramp up, Greene added.
Normally, the allowed system is stunningly skillful at recognizing the enemy and ignoring, or tolerating, our ain cells. Sometimes, however, this procedure goes awry. That's when T-cells (another type of white blood cells) come up in. The trunk uses these T-cells to cantankerous-check targets — only if both a B-cell and a T-cell recognize something as a foreign invader volition an immune response be triggered, Goodman said. The body is supposed to remove B-cells that make so-called machine-antibodies, which react to the body's own cells. But when that doesn't happen, the body may marking its own cells for destruction and then relentlessly eliminate them. Autoimmune diseases such every bit lupus, rheumatoid arthritis, or type i diabetes tin effect, Goodman said. There are more than 100 autoimmune disorders, according to the American Autoimmune Related Diseases Association.
What are monoclonal antibodies?
Antibodies have get the basis for some of the virtually useful medicines, besides as some of the about powerful lab techniques in biology, Goodman said. Ane of these clinical and therapeutic superstars is what's known every bit a monoclonal antibody.
To create a monoclonal antibody, researchers vaccinate an creature (or possibly a human) to stimulate the production of antibodies against a detail substance. The body will gradually make antibodies that are more than and more constructive against that antigen. These antibody-producing cells are then filtered out of white blood cells and put into a dish to run across which cells bind the antigen all-time, Goodman said. The cell that binds the best is then isolated — it is an antibody-producing factory, specifically honed to churn out i super-selective antibody.
From there, that cell is fused to a blood cancer jail cell, producing something chosen a hybridoma. This hybridoma, or monoclone, is an inexhaustible generator of exactly the same antibody, over and over and over. (Researchers bind the monoclonal prison cell to a cancer prison cell because cancer just continues to reproduce.)
"It just produces and produces and produces, and it will never stop, and it'due south a cancer, then it's essentially immortal," Goodman said. What it produces is a monoclonal antibiotic.
Related: Ebola survivor's blood could lead to time to come vaccine
Such cell lines have an incredibly diverse range of uses. At that place are millions of commercial monoclonal antibodies, which are used in labs to tag the tiniest, most specific cellular targets for study, Goodman said.
"They're incredible, they're amazingly precise tools," Goodman said.
Monoclonal antibodies also grade the ground for many blockbuster drugs. For instance, the drug adalimumab (brand name Humira), is a monoclonal antibody that treats rheumatoid arthritis by inhibiting an inflammatory protein known as a cytokine. Another, chosen bevacizumab (Avastin), targets a molecule that fuels claret vessel growth; by blocking this molecule, bevacizumab tin tiresome the growth of lung, colon, kidney and some brain cancers.
And in the SARS-CoV-2 pandemic, doctors around the world are racing to create monoclonal antibodies that will hopefully neutralize the new coronavirus, Greene said. These antibodies are filtered from the plasma of people who have recovered from COVID-19 (besides called convalescent serum). The hope is that by isolating the most effective antibodies, then producing them en-masse, doctors can create a treatment that provides a temporary, "passive" immunity until the body tin catch upwardly and mount an effective, more long-lasting response on its own, Greene said.
Related: one in five people tested in New York accept antibodies to the coronavirus
By contrast, polyclonal antibodies are derived from multiple B-cells. Polyclonal antibodies are a library of antibodies that all bind to slightly different parts of the antigen, or target. Polyclonal antibodies are typically produced by injecting an creature with the antigen, stimulating an immune response, and and so extracting the animals' plasma to produce antibodies en masse, according to a 2005 study in the Institute for Laboratory Animal Research (ILAR) journal.
Dissimilar monoclonal antibodies, which can take upwards to 6 months to produce, polyclonal antibodies can be made in 4 to eight weeks, and crave less technical expertise. In addition, for certain types of tests where yous are trying to detect the antigen, polyclonal antibodies might have a meliorate chance of binding to the target antigen, making them potentially more sensitive. The downside of polyclonal antibodies is that, because each individual animal might produce a different array of antibodies, making polyclonal antibodies that are consistent from batch to batch can be more challenging, and it isn't as easy to have a large supply, according to a 2005 study in the periodical Biotechniques.
How do antibody tests work?
Antibody tests detect whether the body has produced detectable quantities of antibodies to a certain molecule, and tin therefore reveal whether someone has been infected by a specific virus or leaner in the past. Normally, these tests are detecting IgM or IgG, Alive Science previously reported.
For instance, SARS-CoV-2 antibiotic tests typically detect either part or all of the coronavirus' spike protein and tin reveal whether someone has had COVID-19 in the by. Because the torso takes time to ramp up its product of antibodies, people usually merely test positive about 2 weeks after they were starting time exposed to the pathogen, Live Scientific discipline previously reported.
Related: Tin antibody tests tell if y'all're immune to COVID-19?
There are ii mutual types of antibody tests — lateral flow assays and enzyme-linked immunosorbent assay (ELISA) tests. Both involve fixing an antigen to a surface and then detecting whether an antibody binds to that antigen. Unremarkably, a chemic reaction, such as fluorescence or a color-modify, is triggered when the antibiotic binds to the antigen. Lateral flow assays are similar to pee-on-a-stick pregnancy tests; rather than pee, for antibiotic tests, blood or serum is washed over the flat surface, which is usually paper. ELISA tests work on a similar principle, only the tests are conducted in microplates and crave a lab technician, and the results may not read out instantly, Charlotte Sværke Jørgensen, who studies Virus and Microbiological Special Diagnosis Serology at the Statens Serum Institut in Copenhagen, previously told Alive Science in an email.
A good antibody test is 1 that produces few false positives and few false negatives, Live Science previously reported. To ensure that happens, scientists need to "calibrate" their test, for example, by making sure that samples known to not have the antigen do not falsely produce a positive test. For example, with SARs-CoV-2, that would mean testing blood samples from before the pandemic started and making sure no samples come up positive. They also need to take samples that definitely take the antibody in them, and make sure the antibody exam does a good job of detecting those positives.
Additional resource:
- Lookout man a video nearly how antibodies piece of work, produced past the Vaccine Makers Project.
- Read more than about antibody tests from the Centers for Affliction Control and Prevention.
- Read what the manager of the National Institutes of Health, Dr. Francis Collins, has to say about SARS-CoV-two antibodies, as published on the NIH director's web log on June 30, 2020.
Source: https://www.livescience.com/antibodies.html
Belum ada Komentar untuk "Draw a Labelled Diagram of Antibody Molecule"
Posting Komentar